When I saw the tweet, it felt like watching pinball for misleading public health hype – ding! ding! ding! – hitting target…
The HPV Vaccine Should Be Preventing Cervical Cancer: Can We Tell Whether It Actually Is?

The suspense is tough. Will the HPV vaccine reduce cervical cancer and related deaths or won’t it? It should, but this is a disease that takes so long to develop, there was never going to be a quick answer.
In my previous post, I discussed the Cochrane systematic review and meta-analysis of HPV vaccine randomized trials (Arbyn 2018). The trials could test whether the vaccination reduces HPV infection and abnormal cervical cells – which sometimes progress to invasive cancer. But they aren’t big or long enough to prove that this reduction translates into a lower rate of cervical cancer. Working out whether vaccination ultimately reduces cancer won’t just take a long time: it won’t be straightforward, either.
I left the issue of tracking what’s happening at that. But the conversation after my post convinced me I should dig into this, too. First, though, a quick recap on cervical cancer and terminology.
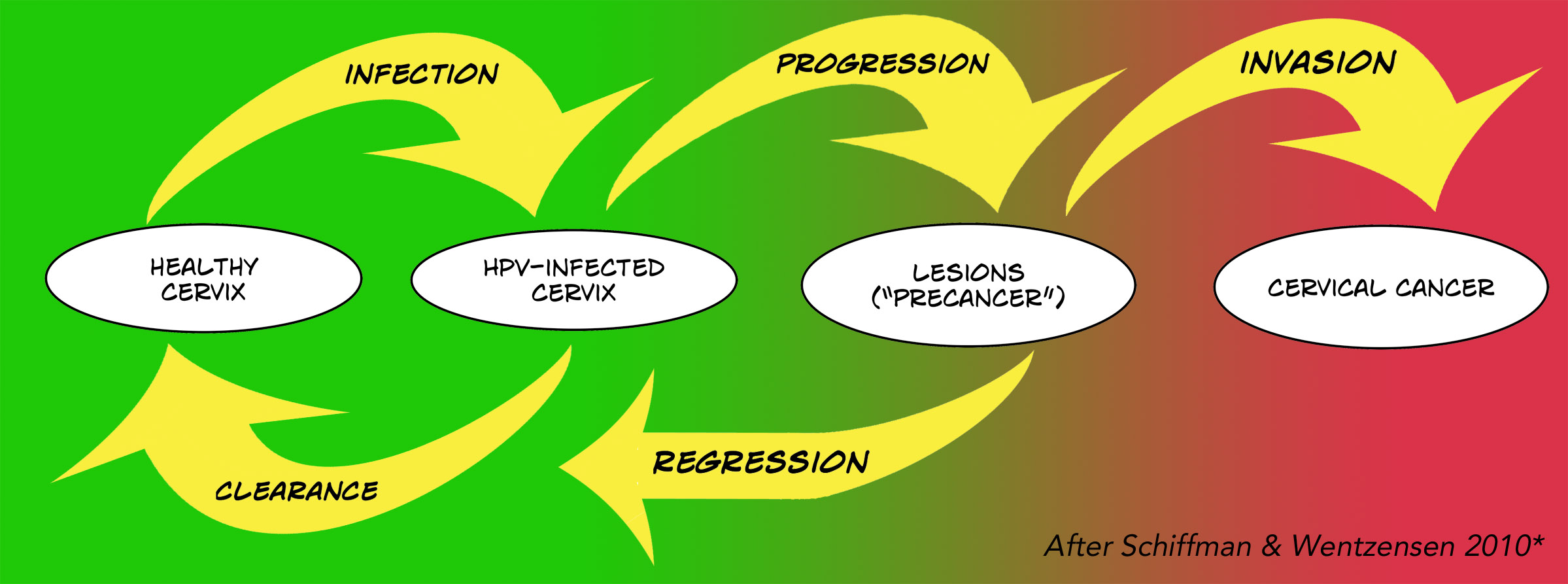
This graphic maps out the complexity of HPV infection and cervical cancer. If you get infected, it’s likely that it will clear up and never cause a life-threatening problem. It can persist though, and cause lesions – CIN (cervical intraepithelial neoplasia) or AIS (adenocarcinoma in situ) – collections of abnormal cells (cytopathology) called “precancer”. But that mostly heals, too, without invading deeper to become cervical cancer.
Just as cancer has stages, cervical lesions have grades. CIN is the most common lesion. When it keeps progressing, this is how it goes: CIN1, CIN2, CIN3, then CIS, which is early stage, non-invasive cancer (carcinoma in situ). Then there are the stages of invasive cancer. The National Cancer Institute (NCI) has an explainer here.
According to an NCI review (Schiffman 2017), about 30% of HPV infections result in the development of abnormal cells:
Most HPV infections clear within 2 years; the 10% that persist for 2 years are highly linked to precancer.
How often does CIN turn into CIS or invasive cancer? I couldn’t find an up-to-date review on this question, and the range of data out there show just how complicated this disease is.
CIN2+ is the level at which treatment is generally recommended, and it’s the measurement used in the systematic review for vaccination. There’s a modeling study that’s useful here. It’s based on data from the cervical cancer screening program Bristol in England, which has a very high rate of participation. The researchers estimated that the rate of progression to invasive cancer is up to 15.4% from CIN2 and CIN3 combined.
There is a review from 1993 that is often relied on, with studies to 1990. The table below includes its conclusions, and some later individual studies found with a quick and dirty search, plus studies cited in major papers. (The sources for my table are listed below the post).
Keep in mind while you look at this, that the studies use different definitions so they are not measuring the same thing. The proportion of women who did and didn’t get treatment varies. The ability to follow women up varies. And the size and risk status of the populations varies a lot, too. Only a very small proportion of these studies followed women for long enough to know how it worked out for them in the end.
|
Lesion Grade |
Invasive Cancer Rate |
|
CIN1 |
1% [a] 0.4% (plus 2.8% CIS) [b] |
|
CIN2 |
5% [a]
1.2% (plus 10.3% to CIS) [b] 0.5% [c] |
|
CIN3 |
12% [a]
3.9% (plus 20.7% CIS) [b] 30% [d] |
| CIS | 30 to 70% [e] |
| Unspecified | 17% (5 of 101 women died) [f] |
What happens once you get the diagnosis of cervical cancer is clear. Cervical cancer has a high mortality rate. There are over 12,000 new cervical cancer diagnoses a year in the US, and over 4,000 deaths from the disease. (37% of women with cervical cancer in the US die within 5 years.) It’s a terrible disease.
What difference could vaccination theoretically make, and when could we know it?
Based on the Cochrane review, the HPV vaccine could reduce the rate of CIN2+ from 6% to 4% in women aged 15 to 26 – but that’s including women for whom the vaccine was too late. Getting vaccinated younger improves the odds of success, because so many young girls won’t already have been exposed to HPV.
The CDC recommends vaccination at 11 to 12 years of age (at the time of other recommended vaccines). The agency first recommended the vaccine for girls in 2006, and for boys in 2011. In 2007, Australia introduced free vaccination in schools for girls aged 12 to 13, and for boys in 2013 – that high level of access makes it an early contender for answers.
The American women who were 12 in 2006 are now only 24 years old. So their risk of having cervical cancer is still very low. Even if a large proportion of them were vaccinated – and very few were back then – we wouldn’t see a big difference for years yet. Per the US Preventive Services Taskforce (USPSTF):
The incidence of invasive cervical cancer peaks among U.S. women aged 40 to 44 years and few cases of cervical cancer are detected in women younger than age 20.

Age-adjusted incidence and death rates of invasive cervical cancer in the United States in 5-year age groups (2000 to 2008). Source: USPSTF.
What could theoretically happen, in ideal circumstances? A 2008 modeling study estimated that preadolescent vaccination could prevent 75% of cervical cancers (range from 60% to 88%), but with 3-yearly screening as well, that could go up to 93% (range from 89% – 97%). In the UK, it’s estimated that there could eventually be a 63% reduction in invasive cancer, with only half as many women having CIN3 before the age of 30 – and impact should be detectable by the time 2019 data from England is available (Cuzick 2010). Another estimate, but with no methodological detail, is 70 – 90% by 2040 (Rahman 2015).
The vaccination rate in the US is far too low to expect anything like that to happen. And vaccination isn’t the only thing that could affect the cervical cancer rate. For example, participation in screening could go up or down, and follow-up could change. Treatments for CIN could improve. People’s behavior could change (sexual, smoking, and more) – and so could the viruses.
The impact of these non-vaccine issues could delay even further how long it takes to get a clear picture, because they could throw up a fair bit of dust. Screening changes have a big impact on diagnoses, and age at diagnosis. Cervical cancer screening will change a lot – when it starts, the time between screens – and HPV testing should detect more disease (and abnormal lesions) than Pap smears could. So there could be spikes or drops that are related to those, not vaccination.
The impact of these kinds of artifactual changes is temporary, but it can seem as though something dramatic is going on with the disease, not the artifact. You can learn the basics about interpreting trend data here at the CDC.

To get an idea of the impact the vaccine could have, we need to have an idea of what the vaccination rate is for young teenage girls in each birth cohort (all the girls born in the same year), and when they start reaching the age where it could change the cervical cancer rate for that cohort.
The mean time from HPV infection to lesions is 3.6 years, and it’s rare to develop cervical cancer in less than 8 years from infection (Cuzick 2010). Exposure to HPV begins with sexual activity. All things considered, people don’t expect to see much change in cervical cancer for a cohort before the women reach 30 years of age.
Globally, only a tiny proportion of women have been vaccinated. By October 2014, an estimated 59 million women worldwide had at least one dose of the vaccine – about 47 million had the full course. That’s well under 2% of women in the target age group – but it’s about a third of those in developed countries. Among the higher income countries (HIC),
successful programs such as those in Scotland and Australia have reached >80% of targeted girls with the full schedule. However, simultaneously, some HIC have struggled to achieve even 50% coverage (e.g. France, USA, Japan, Denmark) due to perceived health concerns fueled by media coverage and/or lack of endorsement from healthcare providers… [Gallagher 2018]
In the US, survey data estimates that the rate of vaccination of girls less than 13 was 14% in 2008, creeping up to 56% by 2012. That won’t be evenly distributed, though. Vaccine coverage is particularly patchy within countries that don’t have a nationwide delivery program through schools. For example, a study of several Swedish counties estimated rates in 2014 for young girls getting more than one dose from 49% to 84%.
The number of women getting cervical cancer before they’re 25 is so low, that the number can fluctuate easily in a smaller country like Scotland (Cuzick 2010). Which is why there’s so much focus on what happens in Australia. As well as being the first country to introduce a nationwide program, and having over 86% of young women with at least one dose of vaccine, the population is 5 times as high as Scotland’s.
The latest available national cervical cancer statistics for Australia, though, are from 2014, and it’s too soon to read much into them. Australia switched to HPV testing in 2017, which is projected to bump up the level of diagnosis of cervical cancers. Taking that and a coverage rate of 82% vaccination for girls and 76% for boys, and a model predicts that the mortality rate from cervical cancer in Australia will start to decline from 2020, with the rate of disease possibly as much as halving by 2035.
There is at least 1 study that has found a statistically significant difference in HPV-related cancer. It’s from Finland (Luostarinen 2018). It’s a long-term cancer registry-based follow-up to the 2,331 Finnish women enrolled in 2 big randomized trials when they were 16-17 years old (FUTURE II in 2002 and PATRICIA in 2004).
At the time, 15,665 unvaccinated 18-19 year-olds who weren’t eligible for the trials were recruited for this study. Later, 6,198 HPV-vaccinated women and 2,173 in a control group of another HPV vaccine trial were added. The cancer registry was searched for cancers in these women in 7-year periods, when they were the same age. They found no cancers in the vaccinated women, but 10 women in the control groups had cancers that are regarded as HPV-related (8 cervical cancers, 1 oral cancer, and 1 vulvar cancer).
That’s important, but it’s just one study – and it’s brief research letter: a full planned study examining the cancers is still to come. And it would be better to analyze at it in the context of all follow-up studies to the trials. There is a lot of data on what’s happening with cervical lesions, though. If there’s any hope of cancer dropping, the rate of moderate and severe cervical lesions (CIN2+) would have reduced by now, certainly in the countries with high vaccination rates. And yes, they have.

Australia first: there was a marked drop in lesions for vaccinated women within 5 years of the vaccination program being implemented (Gertig 2013):
The impact of the vaccine was greatest for women completely vaccinated at younger ages and for the more serious cervical abnormalities CIN3/AIS, which are more likely to be due to the vaccine HPV types.
In the national cervical cancer screening program, the rate of CIN2+ in women under 25 reached “historically low rates” in 2011/2012, according to the Australian Institute of Health & Welfare (AIHW).
The decrease is starting to kick in for older age groups (AIHW):
In those under 20, this decrease commenced from 2007, falling from 11.6 in that year to 3.9 women with high-grade histology per 1,000 women screened in 2016. More recently there was also a decline for women aged 20–24, from 19.7 in 2010 to 10.6 in 2016…
For the first time, in 2014, there was also a decrease in high-grade abnormality detection in women aged 25–29, from 20.3 in 2013 to 18.5 in 2014, a trend which has continued, reaching a detection rate of 15.9 in 2016…There has also been a decrease for women aged 30–34 from 14.1 in 2014, to 13.5 in 2015 and to 12.6 in 2016.
In Scotland, school-based vaccination began in 2008 (a year after Australia). In 2014, a study reported that the rate of CIN2 and CIN3 halved for women who had been fully vaccinated against HPV (from 7.93 per 1,000 to 3.66 per 1,000).
I found 2 systematic reviews of studies trying to tease out whether HPV vaccine is having an impact based on time trend studies (Drolet 2015, Garland 2016). Drolet and colleagues looked at rates of HPV infection and genital warts, not cervical lesions. But Garland and colleagues looked at lesions and cancer, too, although only for the quadrivalent vaccine (which vaccinates against 4 strains of HPV). And they include studies from areas of the US. Drolet found:
[while] vaccination has consistently been successful in reducing HPV 6/11/16/18 infection and disease, reported estimates of impact vary widely… The anticipated benefit of vaccination on HPV-related cancer rates cannot be fully determined yet, because of the long latency periods following exposure.
That’s one of the most critical message from all this, isn’t it? It is definitely too soon to know for sure. The vaccine may have started marketing 12 years ago, but that doesn’t mean you could already expect to see an impact on cervical cancer – especially in a country that had very low uptake of the vaccine back then. And that’s most countries.
The other critical message, though, is that vaccination is changing the trajectory of cervical lesions profoundly in countries with high vaccination rates – and the full extent of that isn’t going to be clear for years, either.
In the Cochrane systematic review, the conclusion was that the vaccine could get close towards halving CIN2/CIN3 in young women. Often, the outcomes in rarefied groups in trials look totally unachievable in everyday life. But this isn’t looking far-fetched at all, in countries providing free vaccination in schools to very young girls and boys.
In those countries, it’s just a few years till the discussion turns to cervical cancer rates, and mortality from it. If/when that happens, I hope we manage to avoid the kind of victim-blaming that started once lung cancer was seen as a kind of personal “choice”. And if it doesn’t happen, I hope we can have a sensible discourse about accepting the disappointment and its consequences.
If a generation of women have lower cancer mortality rates in those countries, the conversation in other countries is going to be painful, though, isn’t it? I’ve had a sinking feeling about this, as my optimism about the HPV vaccine grew this week. There could be giant icebergs of decision regret headed our way.
I hadn’t paid much attention to what happened in Japan when panic about safety of the HPV vaccine flared, but I’ve read about it now. (Here’s an explainer.) It turns out, there was a similar scenario in Denmark, where controversy about adverse effects saw confidence in the vaccine dive: the WHO website says vaccination dropped from 90% of 12-year-old girls in 2014, to below 40%. I guess this victory of fear-mongering is being played out in plenty of other places, too.
I’m not usually a defender of vaccines per se: to me, they’re just a drug class, with each one to be evaluated on its individual merits. I’m one of the people referred to here about the flu vaccine –
So over dinner, I asked a couple of respected evidence-based medicine researchers what they thought of the vaccine. I was taken aback by their response. They basically laughed at the idea that the flu shot was particularly effective.
I sure didn’t expect to be spending so much reading and thinking about the HPV vaccine this month. And I certainly didn’t expect that my Twitter feed would be peppered with people talking about record-breaking measles outbreaks in Europe and Russian bots sowing vaccine discord either!
It’s been a stark reminder though, of why vaccines are in fact different in some ways – and how important community sentiment about them is now. Community sentiment is something we all contribute to, by our words or by our silence. Sure, this is a part of something bigger, that feels overwhelming. But that big thing is made up of all these little ones. And it’s worth getting perspective on them. So many, many bodies and lives are at stake.
Coda:

Here’s a trio of my posts on dealing with phenomena like anti-vaccination partisanship:
- The Skills We All Need to Move Past “Anti-Science” and “Us”
- Post-Truth Antidote: Our Roles in Virtuous Spirals of Trust in Science
- When Science Polarizes: A Personal Activist Story with Evidence
This post is an unplanned part 2 to The HPV Vaccine: A Critique of a Critique of a Meta-Analysis.
~~~~
[Update 1 September 2018] The section on the Luostarinen 2018 study was added after I saw a recent tweet about it by Dan Kegel.
Disclosures: I led the development of a fact sheet and evaluation of evidence on HPV vaccine for consumers in 2009 for Germany’s national evidence agency, the Institute for Quality and Efficiency in Healthcare (IQWiG), where I was the head of the health information department. We based our advice on this 2007 systematic review including 6 trials with 40,323 women, and an assessment of those trials. The findings were similar to those of the 2018 Cochrane review. I have no financial or other professional conflicts of interest in relation to the HPV vaccine. My personal interest in understanding the evidence about the HPV vaccine is as a grandmother (of a boy and a girl).
I am one of the members of the founding group of the Cochrane Collaboration and was the coordinating editor of a Cochrane review group for 7 years, and coordinator of its Consumer Network for many years. I am no longer a member, although I occasionally contribute peer review on methods. I often butt heads with the Cochrane Collaboration (most recently as a co-signatory to this letter in the BMJ). I have butted heads on the subject of bias with authors of the Copenhagen critique.
I worked part-time in a unit of the Australian Institute for Health & Welfare (AIHW) in the 1980s, but in perinatal statistics, not cancer.
26 September: In light of developments, some further disclosures which I did not realize may relate to this controversy and the theories around it, when I first started writing about the Cochrane HPV vaccine review. I was invited to speak at Evidence Live, and my participation was supported by the organizers, a partnership between the BMJ and the Centre for Evidence-Based Medicine (CEBM) at the University of Oxford’s Nuffield Department of Primary Care Health Sciences – the director of the CEBM is the editor of BMJ EBM. Between 2011 and 2018, I worked on PubMed projects at the National Center of Biotechnology Information (NCBI), which is part of the US National Institutes of Health. I am currently working towards a PhD on some factors affecting the validity of systematic reviews.
The cartoons are my own (CC BY-NC-ND license). (More cartoons at Statistically Funny and on Tumblr.)
* The diagram of the development of HPV-related cervical cancer is modeled after one by Schiffman and Wentzensen (2010), via the CDC.
Studies in the table on progression to invasive cancer:
[a] 1993 review, studies with follow-up <1 to 18 years.
[b] “conservatively treated” series (Canada), progression in 10 years.
[c] 2018 systematic review of active surveillance, studies with follow-up 1 to 5 years.
[d] untreated series (NZ) with follow-up after 30+ years.
[e] NCI summary, by 10 to 12 years, no sources provided.
[f] 1978 study of women who did not return for treatment after abnormality detected at screening, cancer rate after a mean of 5 years; 5 of 101 women died of cervical cancer.
Glad I could help 🙂
I have links to a fair number of related studies at http://hpv.kegel.com/faq, http://hpv.kegel.com/safety, and http://hpv.kegel.com/cancer/