Targeting Cancer: A Basketful of Hope
 Targeted treatments for cancer have been extending and saving lives for more than 15 years — precision medicine isn’t a new idea in oncology. Now drugs pioneered on select, specific cancers are, one by one, finding new applications.
Targeted treatments for cancer have been extending and saving lives for more than 15 years — precision medicine isn’t a new idea in oncology. Now drugs pioneered on select, specific cancers are, one by one, finding new applications.
The first wave of targeted drug approvals were for cancers associated with specific mutations. Herceptin (traztuzumab) led the way, approved in 1998. It’s a monoclonal antibody deployed against the HER2/neu receptor that is overabundant in some aggressive and early-onset breast cancers. Robert Bazell’s excellent book Her 2 tells the tale.
In 2001 came the blockbuster Gleevec (imatinib), a small molecule tyrosine kinase inhibitor that intercepts signals to divide. Erin Zammett’s My So-Called Normal Life with Cancer relates that story. A very young editor at Glamour magazine when a routine check-up revealed chronic myelogenous leukemia, Erin’s recovery was one of the first of thousands thanks to this now famous drug.
In 2004 came the monoclonal antibody Avastin (bevacizumab), targeting vascular endothelial growth factor A to squelch the torturous blood supply feeding certain colon tumors. In 2011 Zelboraf (vemurafenib), a serine/threonine kinase inhibitor, entered the market. It blocks signals for melanoma cells to divide in people who have a specific mutation in the BRAF gene called V600. These are just a few.
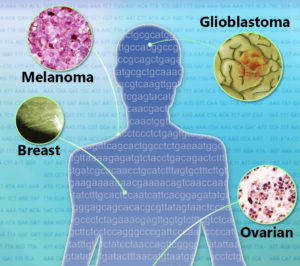
The second wave of targeted cancer treatments began when the trailblazers became available for other cancers. Gleevec is now used for several blood and bone marrow cancers; Herceptin for tumors of the stomach and junction between the esophagus and stomach; and Avastin for types of lung, kidney, ovarian, and brain cancer. A study published two weeks ago in The New England Journal of Medicine reports use of Zelboraf (vemurafenib) for cancers besides melanoma, some quite rare.
The third wave of precision cancer medicines will combine targeted and other therapies. The National Cancer Institute’s Molecular Analysis for Therapy Choice (MATCH) trial will assign patients with lymphomas or solid tumors at 2,400 study sites to receive any of 20 drugs, including combinations, matched to 143 tumor mutations. Much of this matching of medicines to tumors is possible because of The Cancer Genome Atlas (TCGA), an NCI/NHGRI effort to establish mutational landscapes for the more than 200 types of cancer. In 2012, the Pan-Cancer Initiative continued TCGA to systematically explore relationships among tumor locations in the body and mutations.
The recent report on expanding use of the melanoma drug illustrates the insights that arise from these types of investigations. It is an ongoing “basket study” that bundles patients with cancers with targetable mutations that are too rare to fit ongoing clinical trial protocols. The figurative basket includes several types of traditionally-defined cancers. Edward McKenna, Senior Medical Science Director at Genentech, the manufacturer of the melanoma drug, explains. “Mutations targeted by medicines already approved or in development for specific cancers may be present in a broad range of cancers. Often these mutations are only present in a small subset of any specific tumor type. It is not practical or possible to study every medicine and target in every tumor type. Grouping diverse tumors into basket studies allows us to efficiently study them and pursue further evaluation of any promising activity.”
The NEJM study looked at BRAF V600 mutations in colorectal cancer, non–small-cell lung cancer (NSCLC), and the much rarer Erdheim–Chester disease, Langerhans’-cell histiocytosis, cholangiocarcinoma, anaplastic thyroid cancer, and multiple myeloma, as well as a few “orphan” cancers. From 2012 to 2014 the 122 patients from 23 centers worldwide took the pill twice daily.
Q and A

José Baselga, MD, PhD, Physician-in-Chief and Chief Medical Officer at Memorial Sloan Kettering Cancer Center and lead author on the recent NEJM paper, was kind enough to speak with me about the exciting study results.
RL: NBC Nightly News on August 19, when the study was published, showcased a 59-year-old jazz pianist diagnosed with glioblastoma two years previously, whose tumors had vanished following treatment with vemurafenib. Can you describe another notable recovery among the participants?
JB: Yes, The other situation in which treatment with the drug will probably be implemented very soon is Erdheim-Chester disease (a rare form of non-Langerhans cell histiocytosis, it is an adult-onset cancer of tissue macrophages that causes organ failure and bone pain). 50% of patients have the BRAF mutation. This disease has no therapy whatsoever. The first patient we treated had a huge response. She had been on the way to hospice. She couldn’t walk. Two weeks after joining the clinical trial she came back, walking and feeling fine. It’s been over a year since treatment and she is absolutely asymptomatic. We had patients from all over the place coming to be treated on this protocol and every one had tumor shrinkage. None progressed. It was remarkable.
Another great example is non-small cell lung cancer, not only because of the high response rate but because in lung cancer it is common practice to test for EGFR and KRAS mutations, so BRAF should be part of the panel. (Eight of 19 patients with NSCLC had partial responses, median progression-free survival was 7.3 months and is ongoing, and preliminary 12-month overall survival was 66%.)
RL: It must be difficult to recruit volunteers who haven’t been treated, yet that is the optimal way to evaluate a candidate drug. In the study, 89% of the patients had already been treated for their cancers. Will future targeted treatments be given earlier in the clinical trial process?
JB: Patients typically presented at stage 4 and they’d received whatever therapy is approved for their type of cancer, and at that time patients were checked for mutations. I think that as we move on, we will be doing genomic testing up front at the time of diagnosis, and these treatments can become frontline.
RL: David J. Hunter from the Harvard T. H. Chan School of Public Health and Ralph D’Agostino, Sr, from Boston University write in an accompanying Perspective that “the response to a specific mutation is likely to depend on the anatomical site of origin” and that predictions based on mutations “are overstated.” What is the relationship between diagnosis based on location (“conventional tumor nosology”) and diagnosis based on mutation (“molecular nosology”)? Are they both important?

JB: The pendulum has swung. When we began to do studies with gene mutations, testing for the genes we knew and thought were the drivers, there was a school of thought that perhaps the genes and not the tissue of origin would dictate the behavior of the tumor and what therapy would need to be offered to these patients. So people were saying instead of having a breast clinic we’d have a BRAF clinic and a KRAS clinic and a HER2 clinic and so forth. What we’ve learned is that the driver mutation matters, but the tissue of origin is also important. You cannot ignore one or the other; you have to take them together.
RL: Were any of the study results surprising?
JB: Yes, for colon cancer. BRAF colon cancer has a very poor prognosis and median survival of 3 months. We began to treat patients with a single agent (vemurafenib) and had 0% response, not a single patient responded. We were about to close the study and call it a day when two papers were published, one in Nature and one in Cancer Discovery, that said that blocking BRAF activates the EGF receptor. The way to treat these colorectal cancers, then, is to use both a BRAF inhibitor and an EGFR inhibitor. We modified the protocol, adding the EGF receptor antibody cetuximab, and BAM! We saw a tremendous number of responses. We learned that in colon cancer when you block BRAF you also have to block the EGF receptor. This doesn’t happen in lung cancer and in Erdheim–Chester disease.
So before targeted prescribing gets easy it’s going to get complicated, because the tissue-of-origin matters and we will have to deal with both that and the mutation. So our dream of the BRAF clinic goes out the window, and whatever replaces that dream will be complex. These results point to the importance of cell lineage.
RL: TV ads abound for personalized cancer treatment centers, with soothing words and warm fuzzy images. Are such genome-based initial treatments for cancer as available and accessible as these pitches project?
JB: Precision medicine is a buzzword and it has lost some of its value by overuse. Today we have a precision medicine capacity to sequence the genomes of tumors and identify some mutations and based on that information, we might be able to propose tailored treatment. In my opinion that only happens in academic institutions that have research background with serious sequencing efforts.
I think we should stick to the facts. When people announce precision medicine on TV, that’s wrong. I’d never do it. We need to take it seriously and be humble and take a step-by-step approach. At Memorial we are conducting 25 to 30 basket trials, and we offer them to the patients. But this is a work-in-progress. We have a duty to not oversell this to the public.

RL: Where do you see cancer treatment options 10 years from now?
JB: We are going to be able to look at mutations in the blood in a way that will be easy, fast, cheap, and reproducible. We will not only know the mutations, but also how the tumor is evolving to those mutations and the mechanisms for resistance to therapy. We cannot biopsy a tumor every 2 weeks or even months, but we can check the blood as much as we like. That will enable us to use a perfect combination of therapies.
But I don’t think we will cure many patients with just with one therapy. Every tumor has a repertoire of pathways and responses to survive. That repertoire is finite. It has an end. There won’t be a single therapy, so we must be able to combine therapies and identify responses. Then, when things get messy, with multiple mutations far beyond any we could treat, we’ll have the power of immunotherapy. Signatures of genomic alterations predict benefits to some immunotherapies. That is, if a tumor acquires more mutations, there’s a higher likelihood that immunotherapies will identify a tumor as foreign.
Within 10 years, we’ll have the capacity to check more frequently in a readily available fashion. And that is going to enable us to use appropriate combination therapies and also combine immunotherapies and targeted therapies. That’s where we’re going. I’m very hopeful!


[…] Targeted treatments for cancer have been extending and saving lives for more than 15 years — precision medicine isn’t a new idea in oncology. Now drugs pioneered on select, specific cancers are, one by one, finding new applications. The first … Continue reading » […]
Very hopeful news Ricki! Great article!
-Owner of http://www.mazzeodetail.com Premium Automotive Detailing, Bucks County, PA
[…] Targeting Cancer: A Basketful of Hope The second wave of targeted cancer treatments began when the trailblazers became available for other cancers. Gleevec is now used for several blood and bone marrow cancers; Herceptin for tumors of the stomach and junction between the esophagus and … Read more on PLoS Blogs (blog) […]