10 Reasons Why Growing A Human Brain-in-a-Dish is Terrific
 “My brain! It’s my second favorite organ!” cried an aghast Woody Allen as his time-traveling character in 1973’s Sleeper, Miles Monroe, was about to have his head removed at a robot recycling plant. Because my brain is my first favorite organ, I found the news that pea-sized embryonic human brains are growing in a lab at the Austrian Academy of Science, published online in Nature, quite exciting.
“My brain! It’s my second favorite organ!” cried an aghast Woody Allen as his time-traveling character in 1973’s Sleeper, Miles Monroe, was about to have his head removed at a robot recycling plant. Because my brain is my first favorite organ, I found the news that pea-sized embryonic human brains are growing in a lab at the Austrian Academy of Science, published online in Nature, quite exciting.
The “cerebral organoids” take cell culture a giant step forward. These things aren’t just sheets or spheres of identical cells, but organized aggregates of different cell types that seemingly recapitulate a gestating human cerebrum. The organoids are the brainchild of Madeline Lancaster, a postdoctoral researcher in the lab of Jürgen Knoblich.

The fascinating growths unfurl from human induced pluripotent stem (iPS) cells that divide, some differentiating (specializing). Within a month, the cells assemble around a fluid-filled cavity, like summer homes ringing a lake. More accurately, they resemble the neuroepithelial cells that surround a ventricle in the brain – the place where neural stem cells cluster.
To coax iPS cells down the developmental pathway towards being a brain the researchers used special culture conditions, a scaffolding rich in extracellular matrix components, and a spinning bioreactor to bathe the cells in nutrients. Neuroectoderm, the tissue layer from which the central nervous system springs, persists, while other cell types and tissues fade away.
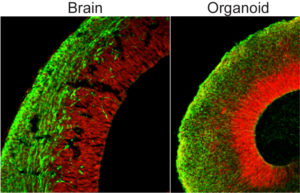
What’s left looks remarkably like smidgeons of the up-to-9-week developing human brain, with neurons firing action potentials, interneurons connecting parts, and stem and progenitor cells keeping the whole thing going. Normal cerebral organoids have discrete regions that resemble different brain parts, such as the ventral forebrain, dorsal cortex, choroid plexus and retina. And cerebral organoids made from a patient with microcephaly reveal exactly what goes wrong to block brain growth.
The news has awakened my inner developmental geneticist, so here’s 10 reasons why I think the cerebral organoids are exciting.
1. Might this milestone temper objections to human stem cell research? Induced pluripotent stem cells, around since 2008, are reprogrammed from skin fibroblasts – no embryos required. In this work and lots of other efforts, the intent was never to grow body parts, at least not now, but to watch development unfold in a way that can’t be done with rodents or cell cultures.
2. An appreciation for self-renewal. The most important characteristic of a stem cell is its ability to self-renew, to make another of itself, for that is what keeps our parts growing as we do. At a stem cell meeting, emphasis is on self-renewal. Yet most popular definitions are so oversimplified as to be wrong: just google “a stem cell turns into any cell type.”
Think about it. If a stem cell turned into any cell type, what would be left to keep churning out new cells, especially if the specialized ones don’t normally divide, like neurons? Self-renewal is the very definition of “stem cell”. It divides to keep itself going. Meanwhile, the second cell from the division may indeed specialize, but typically after further cell divisions, such as the lineages that lead to mature blood cells. Media reports often seem to forget that mitosis yields two cells from one, not one. I can’t wait to see how they handle the cerebral organoid story.
3. Vanishing self-renewal is what goes wrong in microcephaly. Said Lancaster, “The overall size of the patient’s organoids were much smaller than from early stage normal controls. At this point when a neural stem cell population undergoes rounds of division to make more stem cells, patient-derived organoids start making neurons too early. Stem cells not expanding like in these organoids leads to depletion of stem cells, which leads to the overall size decrease. We think this is the basis of the microcephaly in this patient.” Can we please amend the “turn intos” in stem cell reporting?
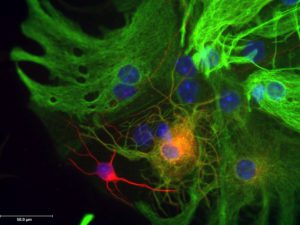
4. Recognition of progenitor cells. When many stem cells self-renew, the other daughter cell isn’t a fully-formed cell type, like a liver or fat cell, but more often a progenitor cell, which is stem-like in that it’s daughters can follow a restricted number of fates,but it can’t self-renew. Progenitor cells are critical go-betweens connecting the stem cells to the neurons in the organoids.
All of this stem cell business is about potential. A stem cell has greater developmental potential – can give rise to more cell types – than a progenitor cell, which in turn has more potential than an immature version of a differentiated cell, such as a reticulocyte that becomes a red blood cell when it extrudes its nucleus. I wrote about using progenitor cells to treat MS just last week.
 5. Organoids bridge the gap between mouse models and cultures of human cells. A mouse falling off a rotating rod just isn’t the same as a person with early-stage Huntington’s disease (HD) sitting in her kitchen crying because she can no longer assemble the ingredients of a meal, or, later on, flailing uncontrollably. Our brains are not quite like those of mice, even those that bear our genes or neural stem cells, and the new work reveals one way that this is so.
5. Organoids bridge the gap between mouse models and cultures of human cells. A mouse falling off a rotating rod just isn’t the same as a person with early-stage Huntington’s disease (HD) sitting in her kitchen crying because she can no longer assemble the ingredients of a meal, or, later on, flailing uncontrollably. Our brains are not quite like those of mice, even those that bear our genes or neural stem cells, and the new work reveals one way that this is so.
Our brains have an outer layer of the subventricular zone that hugs the brains ventricles that mouse brains lack. This might explain our proportionately bigger brains – again, due to self-renewal. Our extra layer harbors types of stem cells and progenitor cells unique to us.
 6. Cerebral organoids make great chapter openers for my textbooks. I had to jettison the example of Phineas Gage, the railway worker who had an iron rod blast through his head and completely change his personality, when evidence emerged that the story had been a tad exaggerated – a little like the subject of two blog posts ago. Out with Gage, in with organoids for the next editions.
6. Cerebral organoids make great chapter openers for my textbooks. I had to jettison the example of Phineas Gage, the railway worker who had an iron rod blast through his head and completely change his personality, when evidence emerged that the story had been a tad exaggerated – a little like the subject of two blog posts ago. Out with Gage, in with organoids for the next editions.
7. Organoids aren’t new, but the new cerebral variety is a great example. The brain bits are in good company: colon, liver, eye and pituitaries. But the brain is more interesting.
8. Applications! Jürgen Knoblich’s excitement came through as he addressed the media yesterday. “Ultimately we’d like to move to more common diseases, like schizophrenia or autism, to study the underlying defects in brain development.”
So many other experiments possible! Addiction. Personality. Tumors. Intelligence. Aggression. Depression. Psychosis. Criminality. And the organoids may help us to understand the beginnings of many brain diseases for which we know genes, but have yet to untangle the exact mechanisms: HD, Parkinson’s, Alzheimer’s, ALS, Rett syndrome, Batten disease, and so many others.
 9. Drug testing. “Organoid culture will make it possible to test drugs directly in a human setting and avoid animal experiments, and get more informed results that can be more easily transferred to cures,” said Dr. Knoblich. DNA Science blog introduced liver organoids for toxicology testing awhile back.
9. Drug testing. “Organoid culture will make it possible to test drugs directly in a human setting and avoid animal experiments, and get more informed results that can be more easily transferred to cures,” said Dr. Knoblich. DNA Science blog introduced liver organoids for toxicology testing awhile back.
10. A philosophical note. “As scientists, we take processes apart. Cortical development entails specification of one area, then progenitor cells generate the correct cell types, and then the cells must migrate to the right positions. All of this we have seen with the organoids at an unprecedented level of precision. But if you zoom out, it’s not a whole brain. We don’t think an organoid system of that kind will be able to recapitulate the organ,” Dr. Knoblich concluded.
 The adult human brain is not simply a conglomeration of the right ingredients, like a cake that arises from the contents of a box plus an egg. Experiences throughout life continually reshape our brains. So even though I won’t be able to get that second brain I’ve always wanted, I’m still excited and encouraged by what the cerebral organoids will be able to tell us about ourselves.
The adult human brain is not simply a conglomeration of the right ingredients, like a cake that arises from the contents of a box plus an egg. Experiences throughout life continually reshape our brains. So even though I won’t be able to get that second brain I’ve always wanted, I’m still excited and encouraged by what the cerebral organoids will be able to tell us about ourselves.
[…] Grow 3-D Human Brain TissuesMIT Technology ReviewABC News -Jersey Evening Post -PLoS Blogs (blog)all 43 news […]
[…] https://blogs.plos.org/dnascience/2013/08/28/10-reasons-why-growing-a-human-brain-in-a-dish-is-terrif… […]
[…] din fizica ale saptamaniiVa spuneam in rubrica de ieri despre creierul fabricat in laborator. Iata 10 purpose pentru care unii sunt excitati de acest proiect…Care e granita dintre iluzie si realitate?Despre ce discutam atunci cand vorbim de genii? Deși […]
[…] Aquí la entrada de blog de Ricki Lewis, en inglés: https://blogs.plos.org/dnascience/2013/08/28/10-reasons-why-growing-a-human-brain-in-a-dish-is-terrif… […]
We are really thankful to Mr.Ricki Lewis for providing his this type of effective information. We hope you will continue your research and give more informative articles.
Thank you. I’m a she.
[…] story from 2013? Atmospheric CO2 hits 400 ppm? Or was it organs from stem cells: liver buds and brain organoids? Or else the host of new gene therapy miracle cures that avoid mentioning AIDS-related vectors? Or […]
I always write that “lentivirus” means HIV when covering gene therapies. But thanks so much for mentioning my organoid blog! I hadn’t thought anyone read that one. 🙂